



|
TOPICS BONOBO Chimpanzee "Ai" Crania photos Itani Jun'ichiro archives Open datasets for behavioral analysis Guidelines for Care and Use of Nonhuman Primates(pdf) Study material catalogue/database Guideline for field research of non-human primates 2019(pdf) Primate Genome DB 
Primate Research Institute, Kyoto University Copyright (c) |
Japanese Generation of macrophages with altered viral sensitivity from genome-edited rhesus macaque iPSCs to model human disease
Iwamoto Y, Seki Y, Taya K, Tanaka M, Iriguchi S, Miyake Y, Nakayama EE, Miura T, Shioda T, Akari H, Takaori-Kondo A, Kaneko S
Abstract
Because of their close biological similarity to humans, non-human primate (NHP) models are very useful for the development of induced pluripotent stem cell (iPSC)-based cell and regenerative organ transplantation therapies. However, knowledge on the establishment, differentiation, and genetic modification of NHP-iPSCs, especially rhesus macaque iPSCs, is limited. We succeeded in establishing iPSCs from the peripheral blood of rhesus macaques (Rh-iPSCs) by combining the Yamanaka reprograming factors and two inhibitors (GSK-3 inhibitor [CHIR 99021] and MEK1/2 inhibitor [PD0325901]) and differentiated the cells into functional macrophages through hematopoietic progenitor cells. To confirm feasibility of the Rh-iPSC-derived macrophages as a platform for bioassays to model diseases, we knocked out TRIM5 gene in Rh-iPSCs by CRISPR-Cas9, which is a species-specific HIV resistance factor. TRIM5 knockout (KO) iPSCs had the same differentiation potential to macrophages as did Rh-iPSCs, but the differentiated macrophages showed a gain of sensitivity to HIV infection in vitro. Our reprogramming, gene editing, and differentiation protocols used to obtain Rh-iPSC-derived macrophages can be applied to other gene mutations, expanding the number of NHP gene therapy models. 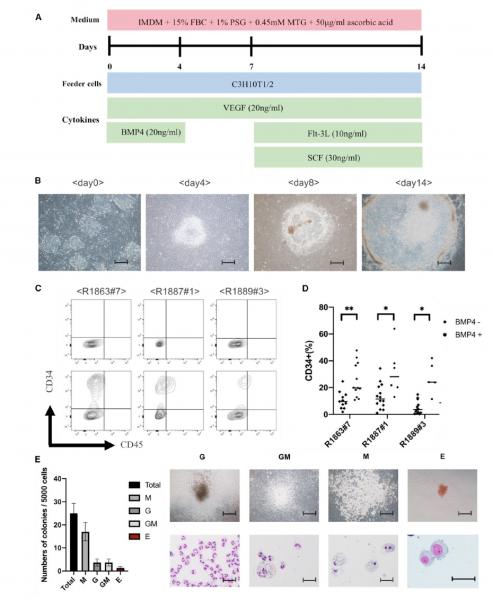 Differentiation of Rh-iPSCs into HPCs. (A) Schematic illustration showing Rh-iPSC differentiation to HPCs. (B) Phase contrast images of Sac differentiation on days 0, 4, 8, and 14. Scale bars, 200 mm. (C) Flow cytometric analysis of the HPC phenotypes 14 days after starting the differentiation. Upper panels, unstained cells; lower panels, stained cells. (D) Dot plots show the differentiation efficiency into CD34+ cells of three Rh-iPSC clones with or without BMP4. *p < 0.05; **p < 0.01. (E) Bar graph displaying the number of colony-forming units. Data are plotted as the mean ± SD of triplicate samples. The microscopic images show colony morphology (upper panels) and cytospins (lower panels). G, granulocytes; M, macrophages; GM, granulocytes/macrophages; E, erythroid. Scale bars, 100 mm (top) and 50 mm (bottom).
Bibliographic information
Molecular Therapy: Methods & Clinical Development 21, 262-273, 2021 DOI: https://doi.org/10.1016/j.omtm.2021.03.008. 2021/03/30 Primate Research Institute
|