A Noncanonical mu-1A-Binding Motif in the N Terminus of HIV-1 Nef Determines Its Ability To Downregulate Major Histocompatibility Complex Class I in T LymphocytesSayuki Iijima, Young-Jung Lee, Hirotaka Ode, Stefan Arold, Nobuyuki Kimura, Masaru Yokoyama, Hironori Sato, Yasuhito Tanaka, Klaus Strebel, and Hirofumi Akari Down-regulation of the major histocompatibility complex class-I (MHC-I) by HIV-1 Nef protein is indispensable for evasion of HIV-1 from protective immunity. Though it has been suggested that the N-terminal region of Nef contributes to the function by associating with a mu-1A subunit of adaptor protein-1, the structural basis of the interaction between Nef and mu-1A remains elusive. We found that a tripartite hydrophobic motif (Trp13/Val16/Met20) in the N-terminus of Nef was required for the MHC-I down-regulation. Importantly, the motif functioned as a non-canonical mu-1A-binding motif for the interaction with the tyrosine motif-binding site of the mu-1A subunit. Our findings will help understanding how HIV-1 evades the antiviral immune response by selectively redirecting the cellular protein trafficking system. Journal of Virology 86, 3944-3951, 2012 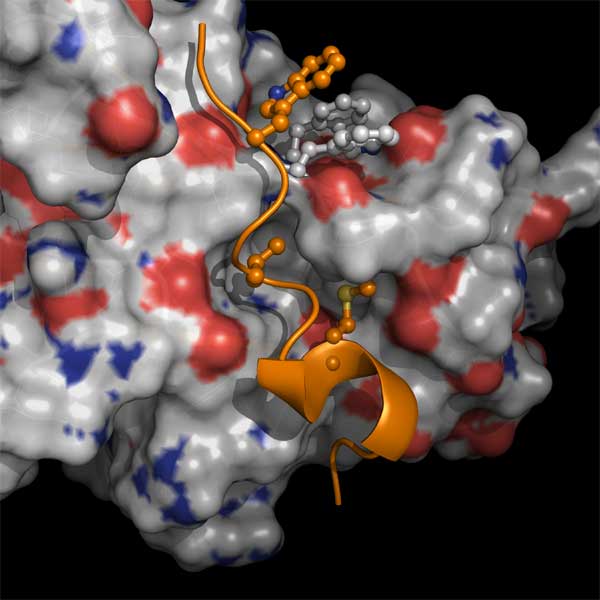
Interaction between a non-canonical mu-1A-binding
motif (Trp13/Val16/Met20) (orange ball and stick representation) of the
N-terminus of human immunodeficiency virus type 1 (HIV-1) Nef and the
tyrosine motif-binding site in a mu-1A subunit of adaptor protein-1
(surface representation). Trp13 in the Nef creates aromatic interactions
with Phe18 and Trp240 in the mu-1A (white ball and sticks), while side
chains of Val16 and Met20 are inserted into a cavity on the surface of the
mu-1A. The interaction is required for down-regulation of the major
histocompatibility complex class-I (MHC-I) and evasion of HIV-1 from
protective immunity. (provided by Dr. Hirotaka Ode)
MAR/13/2012
Copyright(C) 2012 PRI ( ).
All rights reserved. ).
All rights reserved. | 



